Burden of neglected tropical diseases mainly impacts underserved populations

Neglected Tropical Diseases (NTDs) are a group of 20 bacterial, parasitic, viral, and fungal infections identified by the World Health Organization (WHO) that are prevalent in many of the tropical and sub-tropical low and middle-income countries, impacting more than 1.5 billion people6.
These diseases can lead to lifelong disabilities, profound pain and suffering, stigma, isolation and in some cases death. In addition to their impact on health, NTDs contribute to an immense social and economic burden which perpetuates the cycle of poverty by preventing individuals from leading productive lives, and by adversely affecting families, communities, and countries as a whole.
Over 1,7 billion
What Sanofi does...
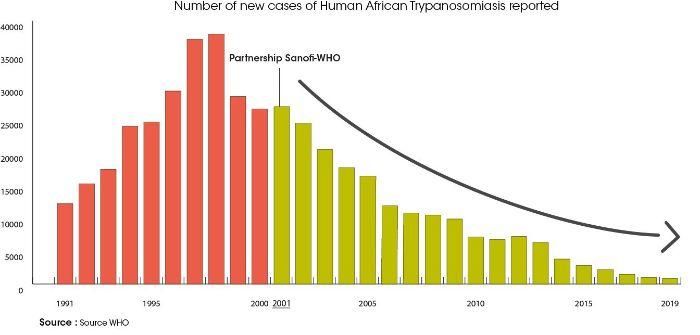
Since 2001 Sanofi has a collaboration program with the WHO in an effort to combat sleeping sickness, leishmaniasis, Chagas disease, Buruli ulcer and yaws. The company has contributed to this partnership with drug donations (such as pentamidine, melarsoprol, eflornithine and today fexinidazole) for treating patients with sleeping sickness or Human African Trypanosomiasis (HAT), and financial support for the execution of programs of the Intensive Disease Management (IDM) department, to an amount of US$5 million per year.
On 30 January 2012 Sanofi, together with other pharmaceutical companies, donors, endemic countries and non-government organizations signed the London Declaration on Neglected Tropical Diseases with the commitment to control, eliminate or eradicate 10 diseases by 2020 and improve the lives of over a billion people, with clear objectives by 2020 for each of these diseases.
Human African Trypanosomiasis (HAT)
Human African Trypanosomiasis, or sleeping sickness, is a vector-borne parasitic disease of sub- Saharan Africa. The WHO with the support of the collaboration program, through the national disease control programs, is able to ensure that all patients with sleeping sickness will receive complex parenteral treatment at no cost.
Through this collaboration the number of HAT cases reported to the WHO has been reduced by ~97% between 2001 and 2019. This is a major achievement considering the logistics challenge it represents.
In 2009, Drug for Neglected Diseases initiative (DNDi) and Sanofi signed a partnership for the development of fexinidazole, a new chemical entity for the treatment of HAT, with DNDi responsible for the clinical development and Sanofi for the industrial development, regulatory submission and supply. The collaboration with DNDi on fexinidazole has made available the first oral monotherapy for sleeping sickness which will allow avoiding systematic lumbar puncture and the need of intravenous infusions and mandatory hospitalization for many patients.
Based on the positive results of the Phase II/III clinical trials completed in the Democratic Republic of Congo (DRC) and Central African Republic, fexinidazole received a positive scientific opinion from the European Medicines Agency (EMA) in November 2018, a decision that paves the way for the distribution of fexinidazole in endemic countries in 2019. On December 24th 2018, a Marketing Authorization has been granted in DRC in gambiense human African trypanosomiasis (HAT).
End December 2019, the first treatments of fexinidazole arrived in Kinshasa (DRC). Through the partnership with the WHO, and in order to support international efforts for eliminating the disease, Sanofi is donating the drug free of charge. The first patients have already started to be treated.
Fexinidazole represents a therapeutic breakthrough which will support sustainable elimination efforts as set out by the WHO’s roadmap for 2020.
Chagas disease
Chagas disease is a parasitic disease transmitted by the triatomine bug, originally from Latin America.
Sanofi, as a partner with the WHO in NTDs Intensive Disease Management, collaborates to achieving the main goals for Chagas disease of the road map for NTDs’ control, elimination or eradication for the period 2012-2020:
- Interruption of intra-domiciliary vectorial transmission in Latin America.
- Interruption of Chagas disease transmission through blood transfusion in Latin America, Europe and Western Pacific
- Elimination of transmission through organ transplantation
- Control of congenital transmission
We contribute to the research of improved solutions, establishing Public-Private Partnerships with national programs improving treatment accessibility/affordability and partnering with academics/patients associations for expanding awareness.
Explore more
Breakthrough on Sleeping Sickness

Sleeping Sickness
Reference
6. https://www.who.int/neglected_diseases/news/treating-more-than-one-billion-people-fifth-consecutive-year/en/, accessed on February 9, 2021
7. http://apps.who.int/gho/data/node.main.A1635?lang=en - Accessed on March 24 2020
8. https://www.who.int/news-room/fact-sheets/detail/trypanosomiasis-human-african-(sleeping-sickness) - Accessed on March 24 2020