Shape Matters: Inside Molecular Clamp Vaccines
Most people don't think about how a vaccine is designed. They think about the appointment, the needle, maybe a sore arm later that day. But behind the scenes, scientists are working through a surprisingly delicate question: to design a more effective vaccine, how do you show the immune system what a virus really looks like?
This question matters more than most of us realize. Consider respiratory syncytial virus (RSV). Most RSV infections resemble the common cold. But globally, RSV triggers 33 million severe respiratory infections in young children each year, with 95% occurring in low- and middle-income countries. And in older adults, RSV is increasingly recognized as a serious cause of hospitalization, with disease severity that can rival influenza in vulnerable populations.
For Keith Chappell, Scientific Advisor at Sanofi, this burden pointed to a challenge he’s been trying to solve for more than a decade: how to capture a moving target.
Vaccine Design and the Shape-Shifting Challenge of Viral Proteins
The immune system doesn't recognize a virus by name. It recognizes shape. On the surface of every virus are proteins that act like tiny grappling hooks, essential for invasion. When a virus binds to a cell, these proteins transform. They shift shape, and that change allows the virus to fuse with the cell, enter it, and begin the infection.
For an RSV vaccine to work well, it must show the immune system what these proteins look like before the shape transformation occurs – what researchers call the “pre-fusion” form of the protein. Train the immune system to recognize that pre-fusion shape of RSV, and the immune system can attempt to stop the virus before the infection.
That’s where the challenge in vaccine development begins.
The viral proteins that provide the best protection against RSV infection are also the least stable. In nature, they exist in a tense, fragile state, like a coiled spring under tension. But once researchers remove these proteins from the virus and produce them in a laboratory, something predictable happens: the proteins relax. They shift into a different, more comfortable shape.
Except that's exactly when researchers, who want to harness those fragile proteins to train the immune system, need them to stay tense.
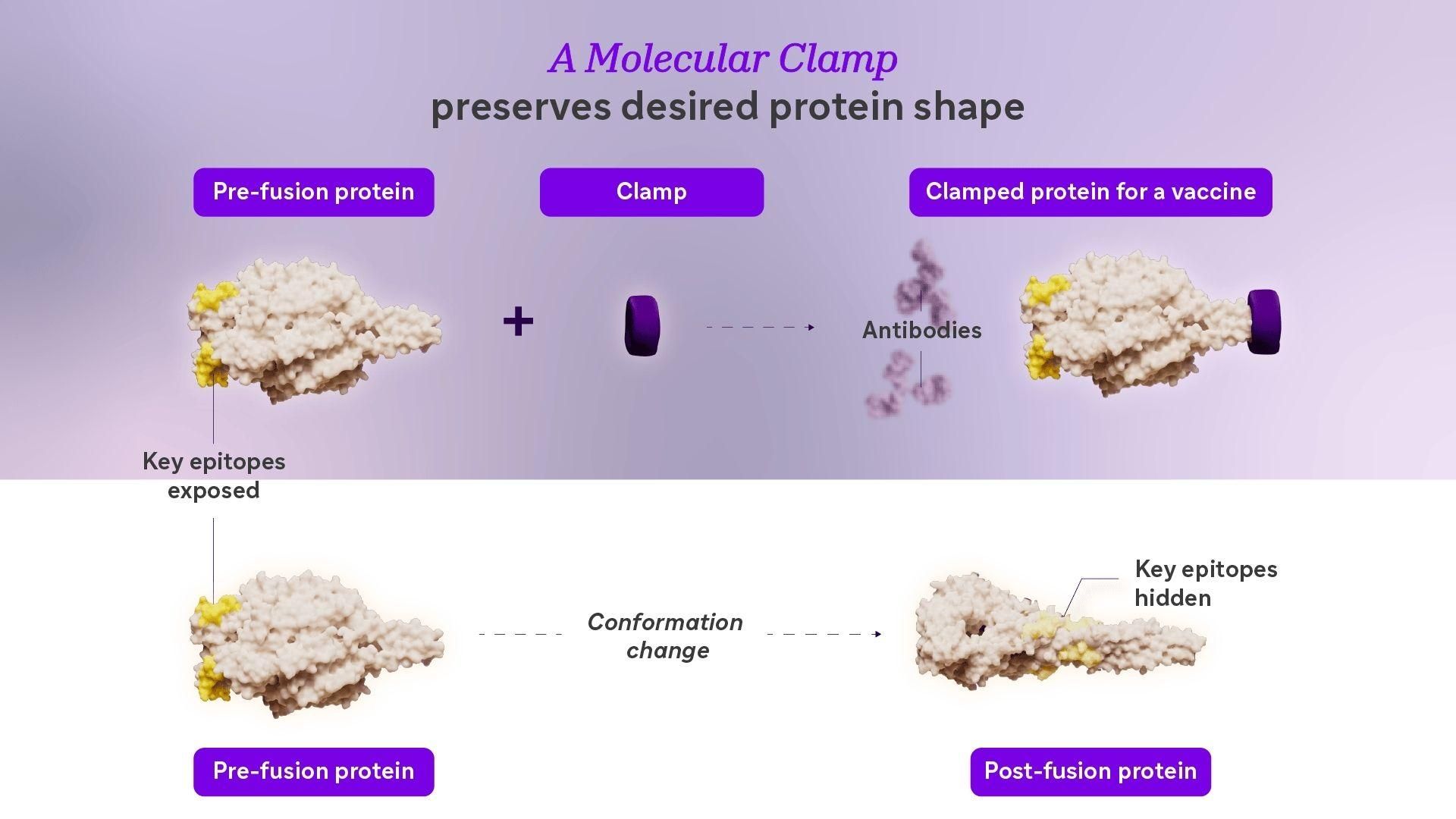
Figure 2. A molecular clamp preserves desired protein shape. When a molecular clamp (purple) is used, the F protein of RSV remains in its pre-fusion conformation (top). The molecular clamp enables the protein’s key epitopes, highlighted in yellow, to remain available for the immune system, and the production of antibodies. Without a molecular clamp (bottom), the F protein of RSV goes through a shape change.
“This shape shifting makes it very difficult to produce the proteins in isolation that are in the correct, unstable structure,” Keith explains. By the time the proteins are purified and formulated during vaccine manufacturing, they may no longer resemble the version the immune system needs to recognize as an infectious threat. The immune system might still respond. But it won’t generate the most protective antibodies, the ones capable of blocking infection at its earliest step.
So, how do you hold something in a shape it naturally wants to leave?
An Insight From Natural Infection: Targeting the Pre-Fusion Viral Protein Shape
The answer began with an observation made by the team Keith was a part of earlier in his career. When patients recovered from natural RSV infection, their bodies produced potent neutralizing antibodies. But those antibodies weren't targeting the virus after it had infected cells. They were targeting the viral protein before it changed shape to enter the cell.
That observation reframed the entire challenge: the pre-fusion structure was the key target. If antibodies recognized the pre-fusion form of the protein, then a vaccine would need to present that structure. The problem now was stability. How do you keep vaccine proteins in their pre-fusion form long enough for the immune system to learn?
The Molecular Clamp Structure: Borrowing From Nature
Rather than redesign the process to produce different proteins, Keith and his team borrowed stability from nature.
They created a “molecular clamp,” a small, synthetic scaffold that holds viral proteins in their unstable, pre-fusion shape. This clamp helps ensure that when a vaccine enters the body, the immune system sees the virus in the shape most associated with strong immune responses.
Keith often describes it using a simple image: Imagine a bouquet of flowers. The flowers are the viral proteins. The clamp is the hand holding the bouquet together. Without the hand in place, the arrangement falls apart.
This scientific work led to the creation of Vicebio in 2018, a biotechnology company dedicated to developing molecular clamp technology. The company is now part of Sanofi.
Then in early 2020, the platform faced its first real-world test.
Vaccine Technology Tested in a Global Crisis
As COVID-19 spread globally, researchers moved quickly to apply the molecular clamp approach to a SARS-CoV-2 vaccine candidate. The urgency was unprecedented. Labs around the world were racing to develop vaccines against a virus that had effectively shut down entire countries.
The program advanced into clinical trials. The vaccine proved safe. It generated an immune response. And then a complication surfaced.
The original molecular clamp design used a structural element borrowed from an HIV protein. Scientifically, the choice made sense; the HIV protein provided excellent stability and posed no infection risk. But in some trial patients, it triggered antibodies that interfered with certain HIV diagnostic tests, creating the possibility of false-positive results. The program stopped. "It was fast in, fast out," Keith recalls. "But we learned a tremendous amount."
For Keith, the setback reinforced something he’s always believed.
Behind every vaccine are people who have devoted their careers to protecting families and preventing severe illness.

Keith Chappell
Scientific Advisor
Rather than abandon the platform, the team redesigned it. A second-generation clamp replaced the HIV-derived element with a different stabilization domain, preserving the structural benefits while eliminating the diagnostic interference. What began as a setback became an engineering lesson. The platform emerged stronger.
Following Vicebio's acquisition by Sanofi, the goal is to bring this work to a broader global stage, combining scientific innovation with experience in large-scale vaccine manufacturing.
A Familiar Manufacturing Process, Designed for Speed
Solving the shape problem was only half the challenge. The molecular clamp also had to be practical and efficient to manufacture at scale.
Fortunately, the manufacturing process is similar to methods already used to produce other biological medicines, like therapeutic antibodies. This allows vaccine makers to use existing infrastructure, rather than building new specialized facilities, supporting reliable production at scale.
But the real advantage lies in speed. Because the clamp structure stays the same across different vaccines, only the genetic sequence of the target viral protein needs to change. Once a pathogen's genome is known – which can happen within days of identifying a new viral threat – researchers can design new vaccine candidates and move quickly into testing.
This plug-and-play approach has particular value for pandemic preparedness. In the event of a new viral outbreak, the ability to rapidly adapt an existing vaccine technology could significantly compress development timelines. Rather than starting vaccine design from scratch, researchers could swap in a genetic sequence from the new pathogen and leverage a validated manufacturing process.
This capability strengthens global biosecurity by reducing the window between identifying a pathogen and delivering a vaccine, saving time in a crisis where every day matters.
Logistics matter, too. Clamp-based protein vaccines are designed for standard refrigeration (4oC), rather than ultra-cold storage (-80oC). This simplifies transportation and storage, particularly in regions where maintaining extremely cold temperatures is difficult or impossible, enabling vaccines to reach more people around the world.
Combination Vaccine Approaches: Toward Simpler Protection
The molecular clamp technology also enables combination vaccines, single shots that protect against multiple respiratory viruses. Combination approaches reduce the number of vaccines needed, and evidence suggests this could increase adherence while saving time and reducing logistical complexity. For patients and healthcare systems, this could mean more disease prevention.
A bivalent vaccine targeting RSV and human metapneumovirus (HMPV) is under evaluation, in addition to a trivalent formulation targeting RSV, HMPV, and parainfluenza virus (PIV3). Combining multiple viral proteins into a single vaccine is scientifically complex; each component must remain stable within the same formulation without interfering with the others. Starting with proteins already stabilized by molecular clamp may help address this challenge.
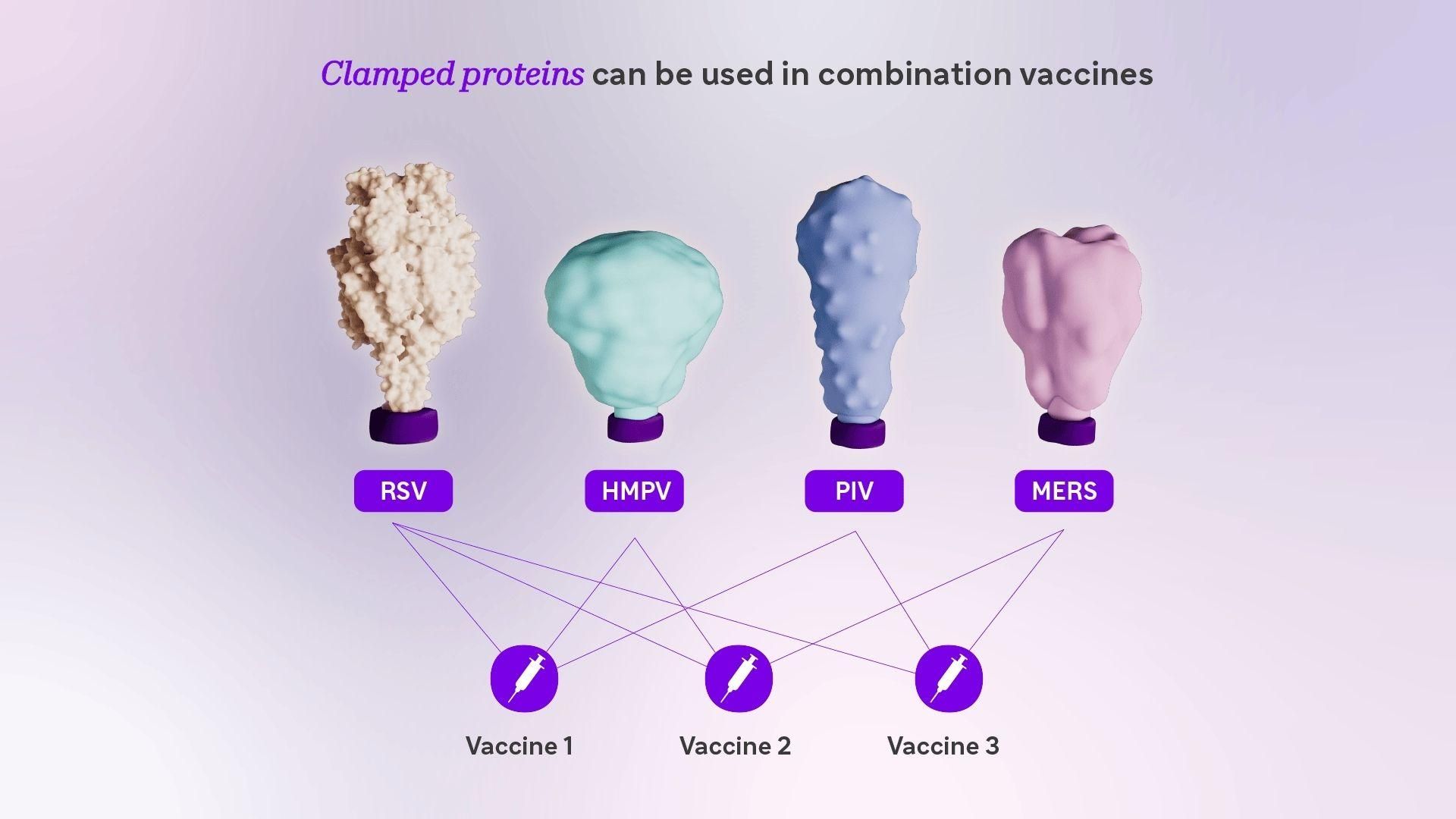
Figure 3. Different proteins are shown with molecular clamps. These proteins can be used in varying combinations to produce new vaccines that target multiple infectious threats.
Looking ahead, combinations might include influenza or COVID-19, potentially offering broad respiratory protection in a single annual vaccination. The long-term goal is to simplify protection, reducing the number of separate vaccinations people need over time, while maintaining or improving efficacy.
Keith emphasizes that the molecular clamp approach is not meant to replace other vaccine technologies. "I think this is absolutely a complement," he says. "Each platform has its pros and cons." Different viruses, different populations, and different contexts may call for different approaches. Expanding the range of tools available to researchers strengthens the ability to respond to evolving health challenges.
Molecular clamp technology started with a simple insight: shape matters, and the immune system must see the right one. If it fulfills its promise, its impact will be measured not only in scientific progress, but in fewer hospitalizations and lives quietly protected from severe disease.
Page updated April 2026