In the Spotlight
Our Science
April 20, 2026
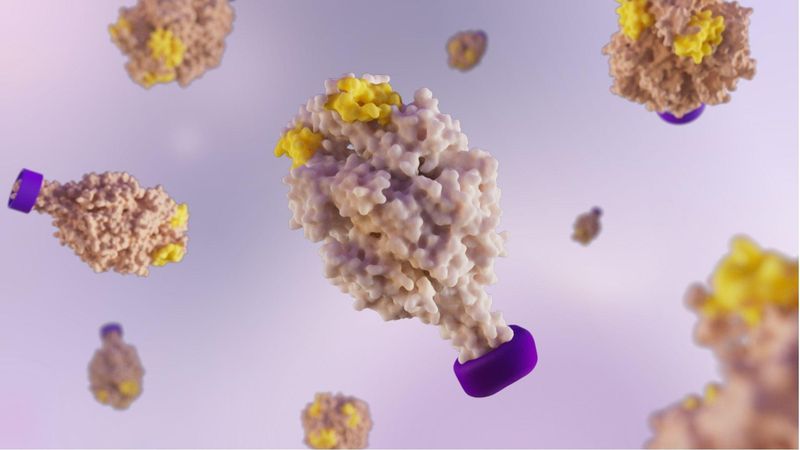
Shape Matters: Inside Molecular Clamp Vaccines
Most people don't think about how a vaccine is designed. They think about the appointment, the needle, maybe a sore arm later that day. But behind the scenes, scientists are working through a surprisingly delicate question: to design a more effective vaccine, how do you show the immune system what a virus really looks like?

Your Health
April 17, 2026
It’s Time to Look Beyond the Bleed
Our Science
January 26, 2026
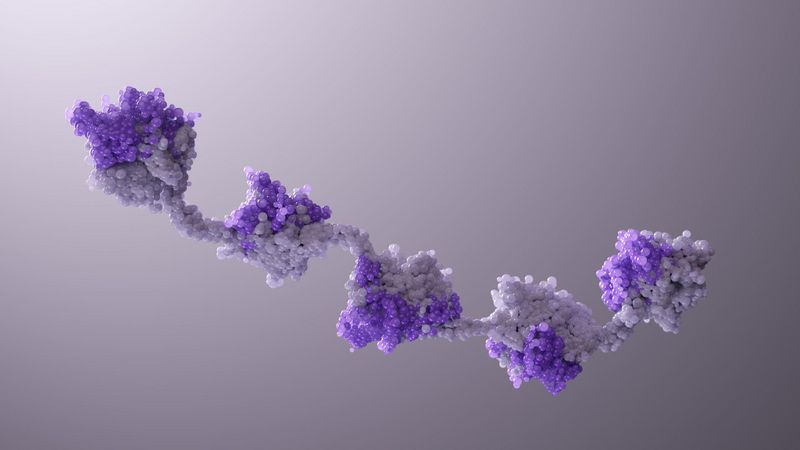
Two’s Company. Three’s a Crowd. Five Nanobody® Subunits? That Could Be a New Medicine.
Watch Our Latest Videos

AI in Manufacturing & Supply

How is AI Speeding up Drug Discovery?

Is AI a Superpower?

The Sanofi Connection
All Stories
Year
Topic
Your Health
June 2, 2026
Charting Progress, Changing Outcomes: A New Era of Multiple Myeloma Care
Sustainability
May 29, 2026
How Sanofi Is Working with Communities While Reducing Its Emissions
Our Science
May 22, 2026
Redefining What’s Possible Through Gastroenterology Research
Our Science
May 20, 2026
Designing Trials That Reflect Real Lives
Our Science
May 20, 2026
Beyond Clinical Trials: Listening Is the Work
Your Health
April 27, 2026
Global Hemophilia Survey Amplifies Patient and Caregiver Voices
Our Science
April 20, 2026
Shape Matters: Inside Molecular Clamp Vaccines
Your Health
April 17, 2026
It’s Time to Look Beyond the Bleed
Our Science
April 3, 2026
