Leading with disruptive technologies and next-generation development platforms enhanced by artificial intelligence (AI), we are building therapeutic platforms that will power our pipeline today and in the future.
Areas of Innovation
Computational Technologies
Every step of our R&D process has undergone a deep digital transformation. We have integrated AI to converge data, profile diseases, and better predict which early molecules have the best potential as medicines. By partnering technology with our deep expertise in immunoscience, we are generating better insights, faster, to accelerate research, streamline development and focus our scientists on solving the problems that matter most to patients.
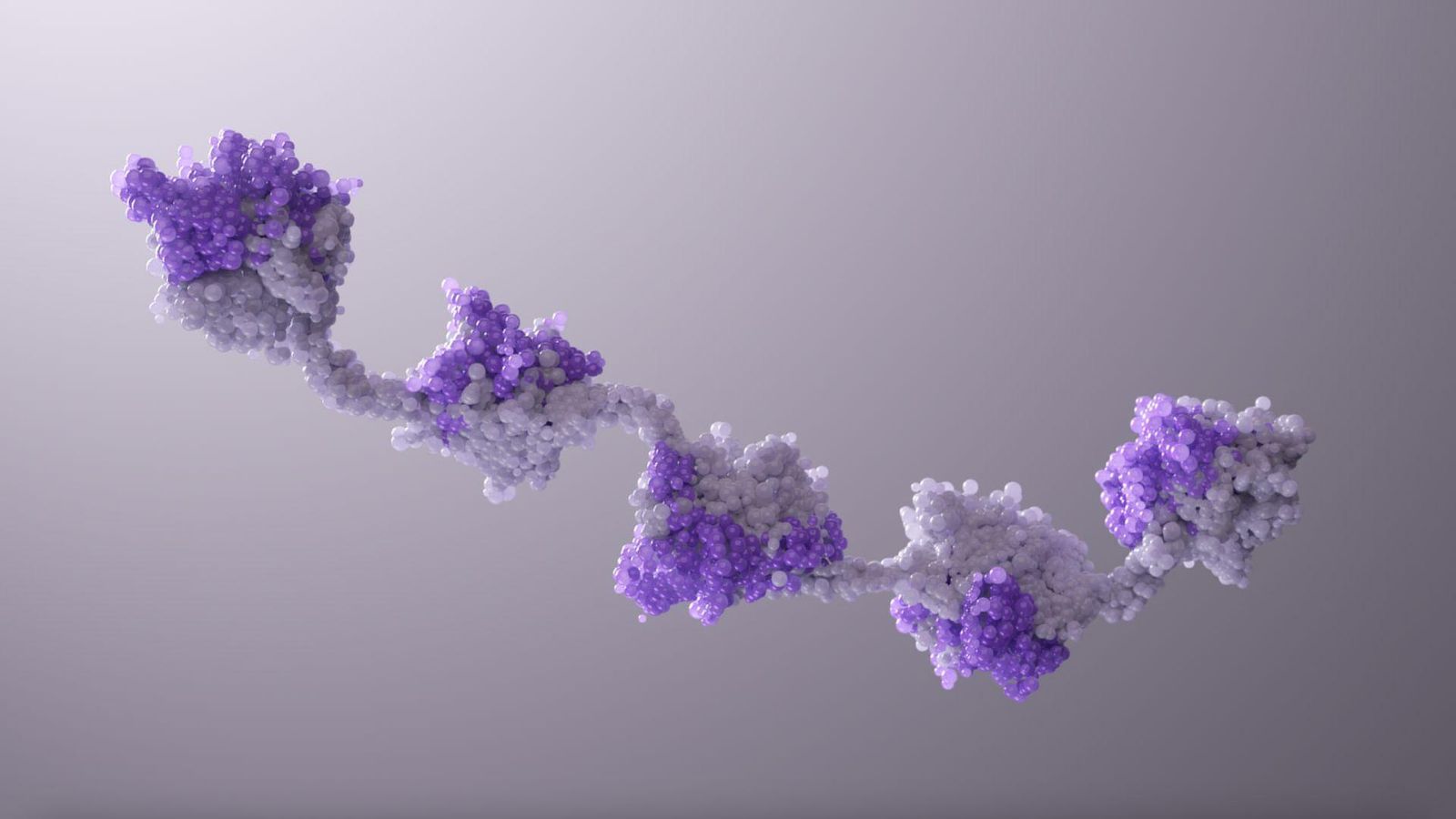
Immune-mediated Technologies
We are putting our decades of expertise into transforming biologic molecules into medicines for neurological diseases, immune-inflammatory conditions and different kinds of cancers. Our robust pipeline includes:
- Multi-specific antibodies, which recognize several targets in a single therapy to tackle a disease on multiple fronts.
- NANOBODY® molecules, designed to reach new, previously inaccessible disease targets and break through efficacy ceilings of existing therapies.
- NK cell engagers, which are intended to help fight cancer without triggering an excessive immune response.
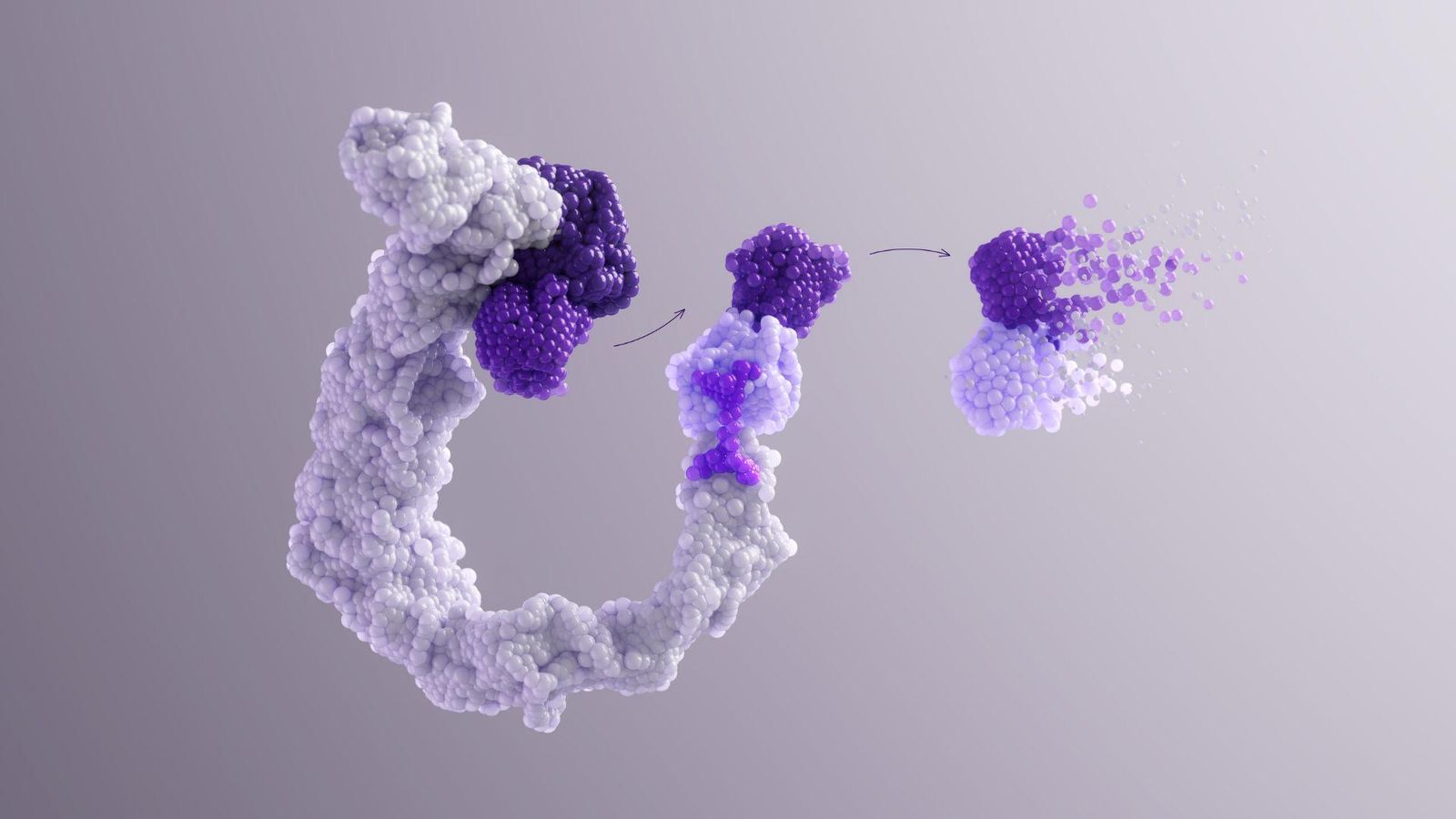
Targeted Protein Degradation
Our deep understanding of cell biology is driving our exploration of the body’s own machinery to break down disease-causing agents within cells. Many diseases are caused by the malfunctioning of specific proteins that are involved in the immune response. By harnessing cells’ natural ability to degrade proteins that are old or damaged and targeting it at these disease-causing proteins, we can potentially stop their activity and alleviate disease.
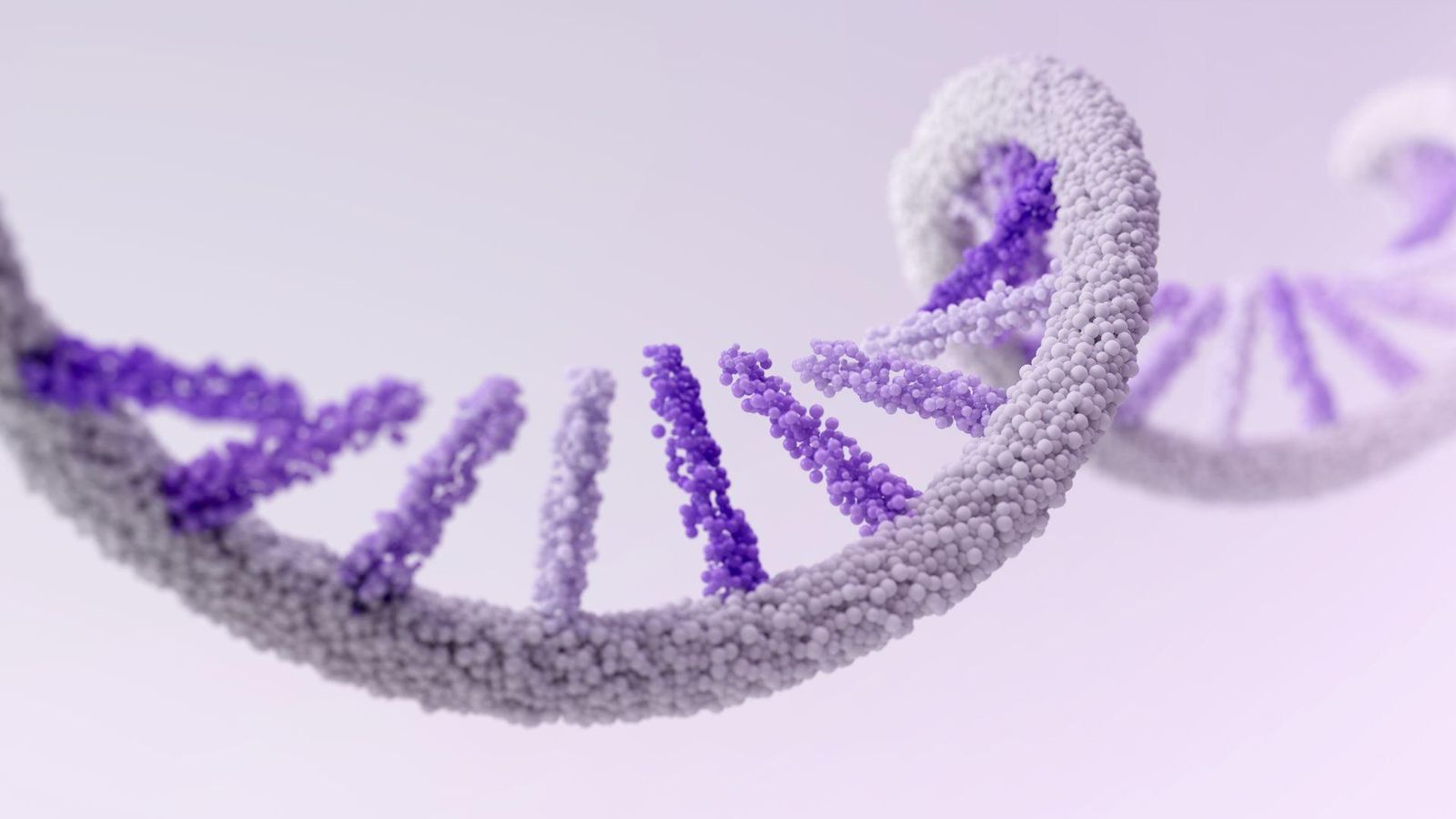
mRNA
We are pioneering the next generation of vaccine and therapeutic platform technology with messenger RNA (mRNA). mRNA technology has the potential to use the body’s natural mechanisms to assemble the proteins it needs to fight disease. These can be proteins which stimulate the immune system to produce antibodies against invaders in the case of vaccines, or proteins that act as therapeutics in the body. We are building on our legacy in vaccines to extend this potential to cancer and hard-to-treat rare diseases.
From Technology to Therapeutic
Learn more about our extensive bank of technology platforms.
Vaccines Beyond mRNA
Our vaccines are as diverse as the diseases they help prevent. Our technologies range from inactivated and live-attenuated vaccines to recombinant protein and polysaccharide-based platforms.
- Live-attenuated vaccines, which are based on weakened whole viruses that can help the body build its defenses without causing disease.
- Recombinant protein vaccines, which are made by producing specific proteins from a pathogen in the lab and formulating them into a vaccine.
Learn More About Our Innovative Technologies