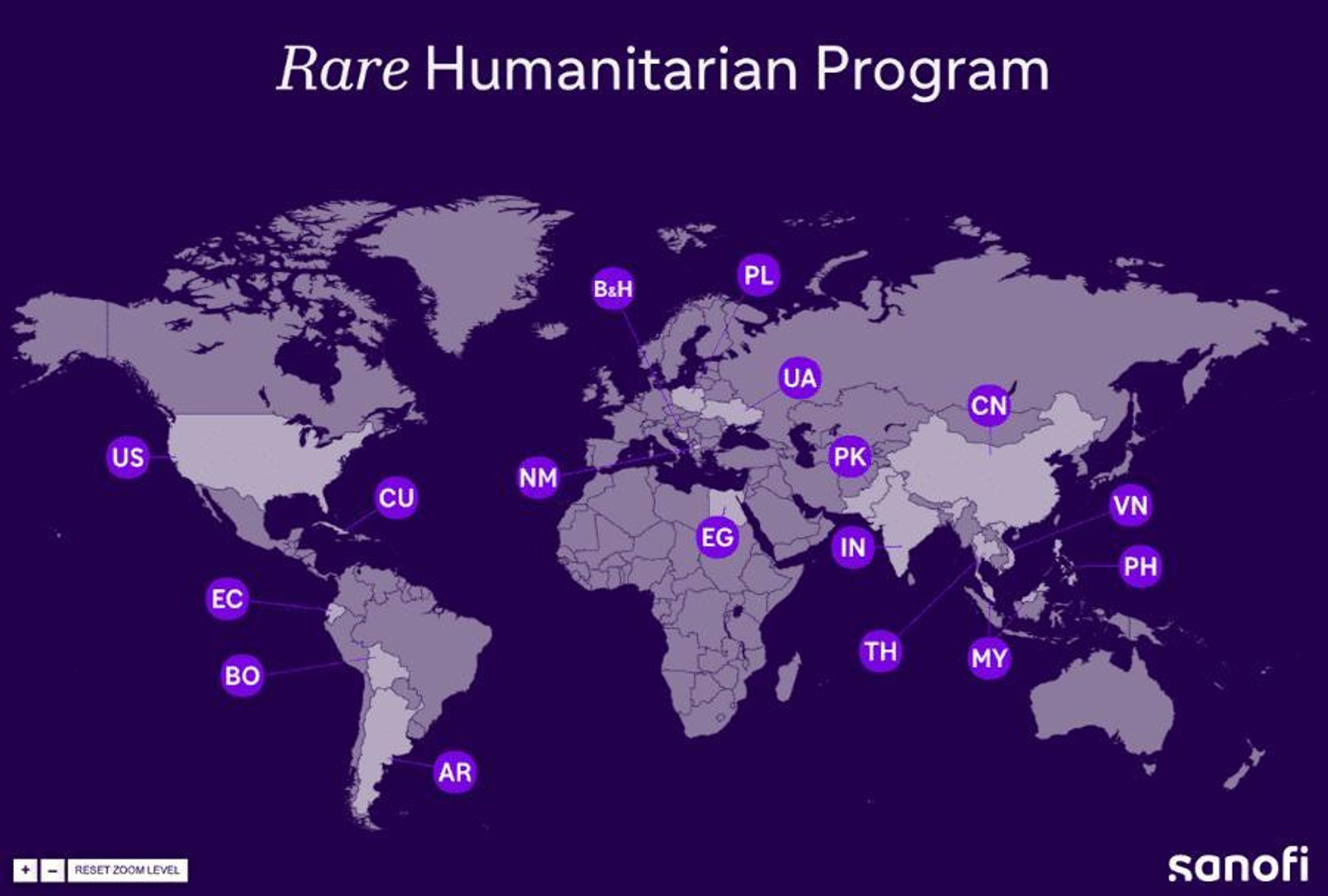
The program provides access to free treatments for patients who meet the program’s criteria and otherwise would not have access to such treatments. We have since expanded support to five additional LSD communities, which includes Fabry disease, Pompe disease, acid sphingomyelinase deficiency (ASMD), and mucopolysaccharidoses (MPS) types I and II.
You can read about the impact of our rare disease humanitarian program, and the lessons learned from three decades of collaboration with local authorities and non-governmental organizations, in the Orphanet Journal of Rare Diseases.1
3,500+
* Internal Sanofi data
Stories of Hope
Since 1991, we’ve helped improve the health and lives of thousands of people with rare diseases around the world. Each of them has a unique story to share.*
* Each patient story reflects the real-life experiences of individuals diagnosed with a specific disease. Individual patients may experience these conditions differently.
For Healthcare Providers
To submit a request for a person living with one of the above listed lysosomal storage disorders, please access the Rare Humanitarian Patient Online Referral Tool (PORT).
Hemophilia Humanitarian Aid
More than 75% of people with hemophilia have limited or no access to diagnosis and treatment. Many people with severe hemophilia in the developing world often do not survive to adulthood, or face a life of disability, isolation, and chronic pain. Sanofi believes all patients should have access to care regardless of where they live.
Sanofi, together with Sobi, is continuing to deliver on their 2014 pledge to donate up to 1 billion IUs of factor therapy over a ten-year period to the World Federation of Hemophilia (WFH) Humanitarian Aid Program. This is the single largest donation of hemophilia factor therapy in history.
Since donations began in 2015, the commitment has had a far-reaching impact. Over 588 million IUs of donated factor therapy have been used to treat over 18,800 people in 41 countries. This has resulted in*:
588m
* All data as of 2015-2021 provided by World Federation of Hemophilia
** International Units

Blood Brothers: Behind the Lens
As a founding member of the humanitarian aid programme WFH, we are committed to deliver a smooth running, sustainable supply of treatments to people living in developing countries who are affected by haemophilia.
Partnerships for Humanitarian Aid
The local expertise and relationships of our partners is essential to making humanitarian aid work for patients. We’re grateful to those who help us identify and reach patients around the world.
Some of the partners we work with on humanitarian efforts include:





