Taking Down Type 2 Inflammatory Disease
Sanofi scientists are working to tame over-active immune responses that drive systemic, chronic diseases.
People with seemingly unrelated diseases can have similar experiences because of one common factor: excessive type 2 inflammation, which in itself is part of a normal immune response to allergens and infections. However, when it goes into overdrive, it can contribute to inflammatory diseases such as asthma, atopic dermatitis (eczema), chronic rhinosinusitis with nasal polyps, eosinophilic esophagitis (a chronic and progressive digestive disease), or even certain environmental allergies.
"Type 2 immunity evolved as protection against venoms, parasites and dangerous environmental exposures, but excessive type 2 inflammation is dangerous and can have a serious impact on quality of life," explained Frank Nestle, Global Head of Research and Chief Scientific Officer at Sanofi. "It's a common culprit behind unpredictable, sometimes life-threatening asthma attacks, the debilitating itch of atopic dermatitis, and the loss of smell and taste that people experience with nasal polyp growth."
Many people living with certain forms of type 2 inflammatory disease use broad-acting immunosuppressants that can provide relief but can also affect immunosurveillance–the body's security system for recognizing pathogens such as bacteria, viruses, or even pre-cancerous and cancerous cells.
Genetic and environmental factors
For scientists working to untangle the exact mechanism of excessive type 2 inflammation, one challenge is the sheer number and complexity of the diseases involved. Environmental and genetic factors are intertwined, and different for each person.
"Even though type 2 inflammation occurs systemically, it can appear in different tissues depending on the patient's own biology and environment. In research, it comes down to pinpointing the exact cell types and pathways involved, then figuring out how to target them without shutting down the whole immune system," said Nestle.
Calming the immune response
Scientists have been mapping many cells and proteins involved in the type 2 immune response at every step to find a route to new treatments. A landmark moment in this exploration was the discovery that cells communicate with each other to activate immune response or inflammation by releasing proteins called interleukins. Specific interleukins activate type 2 inflammation while others may activate other types of inflammation.
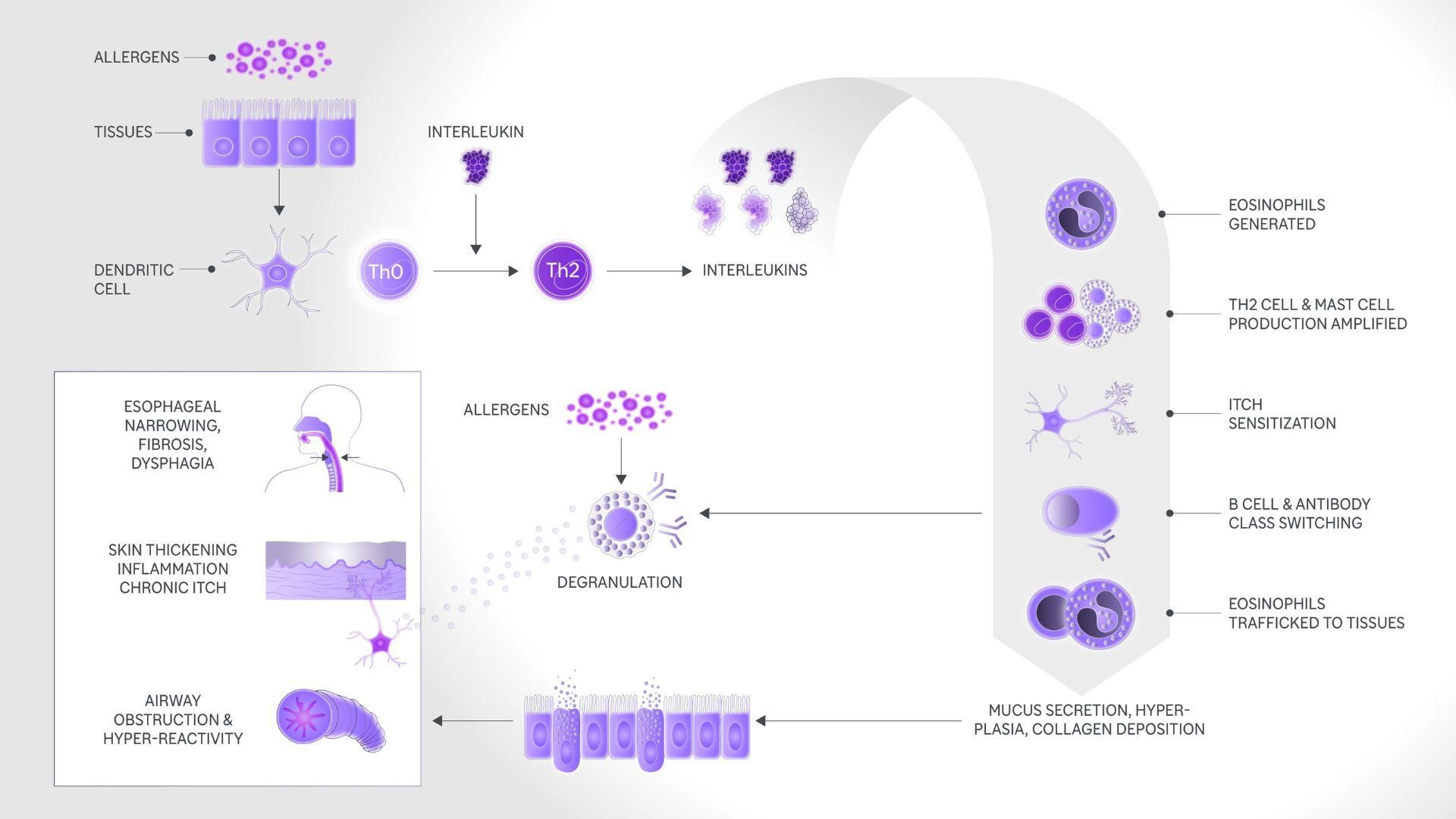
The type 2 inflammation response
Interleukins spring into action when the body's sentinel immune cells (called dendritic cells) detect allergens. In a cascade of biological interactions, they:
- amplify the production and activation of white blood cells;
- activate immune cells called mast cells, which produce histamines and other allergic mediators;
- drive inflammation in specific tissues, for example in the skin and airways;
- cause changes in tissue such as swelling, itching, pain, and mucus production in the airways.
This can trigger a vicious cycle: tissues damaged as a result of these events are prone to infection, which can then cause further inflammation.
Dual pharmacology
Many of the immune cells involved in type 2 inflammation have receptors on their surface that act as docking stations for two specific interleukins. Researchers at Regeneron Pharmaceuticals first observed that these receptors follow a distinct pattern and figured out how to disrupt it. This research led to a new approach: blocking both of these interleukins from binding to their receptors on immune cells.
Type 2 inflammation exists mainly to protect against parasites. By suppressing type 2 inflammation, other types of immune response that fight off bacteria and viruses are still able to function normally.
"We really want to help people who are suffering from excessive type 2 inflammation," said Naimish Patel, Development Therapeutic Area Head for Immunology and Inflammation at Sanofi. "We are focusing our efforts on mounting a two-pronged attack. First, we aim to block immune cells from starting inflammation; second, we need to calm the downstream response that leads to symptoms like itch and wheezing. And we need to try to do that without suppressing the whole immune system."
Inspired immunology
This was a turning point in our type 2 inflammation R&D. It allowed us to explore new frontiers by combining preclinical, clinical, and real-world-evidence studies.
"Sanofi and Regeneron researchers are exploring new solutions for these diseases, leveraging a broad range of biological and clinical expertise, innovative partnerships, and an outstanding toolbox of technologies," said Paul Rowe MD, ATSF, Head of Global Medical, Immunology at Sanofi. "We are trying to get right to the core of what's driving this excessive inflammation so that one day, we can make a lasting difference for patients by determining ways to prevent and treat many debilitating diseases with underlying type 2 inflammation."
Explore more

The Type 2 Inflammation Connection
References
- Gandhi NA, Bennett BL, Graham NM, et al. Targeting key proximal drivers of type 2 inflammation in disease. Nature reviews. Drug Discovery. 2016 Jan;15(1):35-50. DOI: 10.1038/nrd4624
- Gandhi NA, Pirozzi G, Graham NMH. Commonality of the IL-4/IL-13 pathway in atopic diseases. Expert Review of Clinical Immunology. 2017 May;13(5):425-437. DOI: 10.1080/1744666x.2017.1298443
- Nair MG, Guild KJ, Artis D. Novel effector molecules in type 2 inflammation: lessons drawn from helminth infection and allergy. Journal of Immunology (Baltimore, Md. : 1950). 2006 Aug;177(3):1393-1399. DOI: 10.4049/jimmunol.177.3.1393
- Webb LM, Tait Wojno ED. Notch Signaling Orchestrates Helminth-Induced Type 2 Inflammation. Trends in Immunology. 2019 Jun;40(6):538-552. DOI: 10.1016/j.it.2019.04.003